
GlaxoSmithKline
GlaxoSmithKline and Pfizer to create a new world-leading, specialist HIV company
GlaxoSmithKline and Stiefel to create new world-leading specialist dermatology business
āOāēāNā\üEāXā~āXāNāēāCāōüAŖžŹæé╠ōīł¤É╗¢“é╔9.9üōÅoÄæ
http://www.gsk.com/about/history.htm
| ü@ | ü@ | ü@ | ü@ |
| 1830
John K Smith opens his first drugstore in Philadelphia. John's younger brother, George, joins him in 1841 to form John K Smith & Co. |
1842
Thomas Beecham launches the Beecham's Pills laxative business in England. The laxative is to become widely successful. |
ü@ | ü@ |
| 1865
Mahlon Kline joins Smith and Shoemaker - as John K Smith and Co had become - as a bookkeeper. |
1859 Beecham opens the world's first factory to be built solely for making medicines at St Helens in England. |
1873
Joseph Nathan, who left the UK to seek new business opportunities 20 years before, establishes a general trading company at Wellington in New Zealand - Joseph Nathan and Co - the foundation for the Glaxo company to be formed later. |
1880
Burroughs Wellcome & Company is established in London by American pharmacists Henry Wellcome and Silas Burroughs, four years after Joseph Nathan opened a London office. |
| 1875
Mahlon Kline took on additional responsibilities as a salesman and added many new and large accounts. He is rewarded when the company, Mahlon K Smith and Company, is renamed Smith, Kline and Company. |
1885
Thomas Beecham's company acquires headquarters on the corner of Silver Street and Water Street, St Helens, England. Two years later, the company's new factory in St Helens becomes the first in the area to have electricity. |
ü@ | 1884 Tabloid is registered as a Burroughs Wellcome and Company trademark to describe its compressed tablets. |
| 1891
Smith, Kline and Company acquires French, Richards and Company, providing a greater portfolio of consumer brands. |
ü@ | 1906
Glaxo is registered by Joseph Nathan and Co as a trademark for dried milk. |
1902
The Wellcome Tropical Research Laboratories open. |
| 1929 Smith, Kline and French Company is renamed Smith Kline and French Laboratories and becomes more focused on research. | ü@ | 1947
Glaxo Laboratories Ltd absorbs the Joseph Nathan company
and becomes the parent company. Glaxo is listed on the
London Stock Exchange. |
1936 Sir Henry Wellcome's will leaves sole ownership of The Wellcome Foundation Ltd to a UK medical research charity, today called the Wellcome Trust. Sir Henry Dale of Wellcome is awarded the Nobel Prize for Medicine for his work in the chemical transmission of nerve impulses. |
| ü@ | ü@ | 1958
Glaxo acquires Allen and
Hanburys Ltd. |
ü@ |
| 1982 SmithKline acquires Allergan, an eye and skincare business, and merges with Beckman Instruments Inc, a company specialising in diagnostics and measurement instruments and supplies. The company is renamed SmithKline Beckman. | ü@ | 1978 Through the acquisition of Meyer Laboratories Inc, Glaxoüfs business in the US is started, to become Glaxo Inc from 1980. The broad-spectrum injectable antibiotic Zinacef (cefuroxime) is introduced by Glaxo. | ü@ |
| 1989 SmithKline Beckman and The Beecham Group plc merge to form SmithKline Beecham plc. | 1995 Glaxo and Wellcome merge to form Glaxo Wellcome. | ||
| 2000 GlaxoSmithKline formed through the merger of Glaxo Wellcome and SmithKline Beecham. | |||
April 16, 2009 GlaxoSmithKline
GlaxoSmithKline and Pfizer announce innovative agreement to create a new world-leading, specialist HIV company
GlaxoSmithKline plc (GSK) and Pfizer Inc (PFE) today announced they have entered into an agreement to create a new world-leading HIV company focused solely on research, development and commercialisation of HIV medicines. The new HIV business will be more sustainable and broader in scope than either company's individually, and will hold a 19% share of the growing market and have an industry-leading pipeline. GSK will initially hold an 85% equity interest in the new company and Pfizer will hold 15%.
Focused new business with
industry-leading HIV pipeline
The new company will have a broad product portfolio of 11
marketed products
including market-leading therapies such as Combivir, Kivexa and
Selzentry/Celsentri. Based on 2008 pro-forma results, this
combined portfolio generated sales of
approximately £1.6 billion and these revenues will provide
the new company with financial stability and support investment
in its pipeline.
The clear focus of the new business will be to invest in research and development of innovative HIV treatments and formulations that improve adherence and overcome resistance to the virus.
The company will have an industry-leading pipeline of 6 innovative and targeted medicines, including 4 compounds in phase II development. Altogether, the new company will have 17 molecules at its disposal to develop in fixed-dose combinations as possible new HIV treatments. The new company will contract R&D services directly from GSK and Pfizer to develop these medicines.
ō·¢{īoŹŽÉVĢĘ
ÉVē’Äąé═égéhéuŖųśA¢“é┼īv11É╗Ģiéé╗éļé”üAöäÅŃŹéé═24ēŁāhāŗüi¢±2400ēŁē~üjé╔é╠é┌éķüBéQāPā^ɼÆĘé¬ī®Ź×é▀éķŹRégéhéu¢“ÄsÅĻé┼é═āVāFāA19üōéł¼éķæÕÄĶéŲé╚éķüBÉV¢“ī¾ĢŌĢ©Ä┐éÓ6Ģi¢┌éĀéĶüAéżé┐4Ģi¢┌é¬ŖJöŁÆåŖ·é╠ÆiŖKé╔éĀéķüB
éféréjé═ÄÕŚ═é╠ŹRāEāCāŗāXÄ¢ŗŲéŗŁē╗üBātā@āCāUü[é═ō»ŗŲé╠Ģ─āÅāCāXöāĹé┼ĢŌŗŁéĄéĮāoāCāIłŃ¢“Ģié╠ŖJöŁé╔ÅWÆåéĘéķüB
GlaxoSmithKline and
Stiefel to create new world-leading specialist dermatology
business ü@öńĢåē╚
ü@Stiefel, the
worldüfs largest independent dermatology
company, acquired by GSK in deal valued at up to $3.6 billion
ü@New
global business will have combined revenues of approximately $1.5
billion and robust new product pipeline
ü@Significant
step forward to grow and diversify GSKüfs business, providing immediate
new revenue and synergy opportunities
GlaxoSmithKline plc (GSK) and Stiefel Laboratories Inc. today
announced that they have signed an agreement to create a new
world-leading specialist dermatology business. Under the terms of
the agreement GSK will acquire the total share capital of Stiefel
for a cash consideration of $2.9 billion. GSK also expects to
assume $0.4 billion of net debt upon closing. A potential further
$0.3 billion cash payment is contingent on future performance.
GSKüfs existing prescription
dermatological products will be combined with Stiefelüfs and the new specialist global
business will operate under the Stiefel identity within the GSK
Group.
ü@É╗¢“æÕÄĶé╠āOāēāNā\üEāXā~āXāNāēāCāōüiGSKüjé═10ō·üAŖžŹæé╠ōīł¤É╗¢“é╔¢±1300ēŁāEāHāōüi¢±105ēŁē~üjéōŖÄæéĄüAō»ÄąŖö9.9üōéĵōŠéĘéķéŲéŲéÓé╔üAēcŗŲ¢įéŗżŚLéĘéķōÓŚeé╠ÉĒŚ¬ōIƱīgŖųīWéīŗéįé▒éŲé┼ŹćłėéĄéĮüBGSKé═ōīł¤É╗¢“é╠ÉVŖö117¢£ŖöéĵōŠéĘéķüB
ü@ōīł¤É╗¢“é═ŹöNüAöäÅŃŹé8010ēŁāEāHāōüi¢±649ēŁē~üjéŗLś^éĄéĮŖžŹæÉ╗¢“ŗŲŖEŹ┼æÕÄĶüBēpŹæīné╠GSKé═ŹöNüAŖOÄæīnÉ╗¢“ē’ÄąéŲéĄé─é═ŖžŹæÄsÅĻé┼Ź┼Źéé╠4342ēŁāEāHāōüi¢±352ēŁē~üjéöäéĶÅŃé░éĮüBÉóŖEæSæ╠é┼é╠öäÅŃŹéé═ ŹöNÄ×ō_é┼284ēŁā|āōāhüi¢±3Æø9350ēŁē~üjé┼ÉóŖE5ł╩üB
ü@ŗŲŖEŖųīWÄęé═üuGSKé¬Ģ█ŚLéĘéķÉóŖEōIé╚ÉV¢“éŲōīł¤É╗¢“é╠ŹæōÓēcŗŲ¢įé╠ægé▌ŹćéĒé╣é═ögŗyī°ē╩é¬æÕé½éóüBāēāCāoāŗé╠Ŗžö³¢“Ģié¬ōīł¤É╗¢“é╠ŖöÄ«é 8.6üōĢ█ŚLéĄé─éóéķéĮé▀üAōGæ╬ōIöāĹé╠ē┬ö\ɽéÓé│é│éŌé®éĻéĮé¬üAƱīgé╔éµéĶé╗éżéĄéĮī£öOéÓÄŃé▄éķüvéŲÄwōEéĄéĮüB
2010/5/5ü@Æ®æNō·Ģ±
ōīł¤É╗¢“üAÄOÉńŚóÉ╗¢“éöāĹéų
ü@ŖžŹæÉ╗¢“æÕÄĶé╠ōīł¤É╗¢“é═4ō·üAÆåīśÉ╗¢“ē’Äąé╠ÄOÉńŚóÉ╗¢“éŗzĹŹćĢ╣éĘéķé▒éŲé┼é▒é╠é┘éŪŹćłėé╔ÆBéĄüAöāĹŖzé╚éŪŗlé▀é╠īÅ┬éÉié▀é─éóéķé▒éŲé ¢Šéńé®é╔éĄéĮüBöāĹŖzé═500ēŁāEāHāōüi¢±42ēŁē~üjæOīŃéŲé│éĻüAŹūĢöé╠ÄæÄYĢ]ē┐éŹsé┴éĮÅŃé┼üAŹĪÅTÆåé╔éÓÉ│Ä«öŁĢ\éĘéķüB
ü@ÄOÉńŚóÉ╗¢“é╠ŹöNé╠öäÅŃŹéé═361ēŁāEāHāōüi¢±31ēŁē~üjé┼üAāGāCāYÄĪ Ś├¢“é╠ī┤Ś┐é╚éŪéÉČÄYéĄé─éóéķüBō»Äąé═Ź┼ŗ▀üAĢ─ÉHĢiłŃ¢“ĢiŗŪüiFDAüjé®éńÉČÄYÉ▌ö§é╠öFÅžéľé»éĮüB
ü@öāĹé╔é═ōīł¤É╗¢“üAŚ╬Å\ÄÜüAŖžōŲ¢“Ģié╚éŪé¬ŖųÉSéÄ”éĄé─éóéĮüBōīł¤É╗¢“é╔éµéķöāĹé╔é═üAōlÄRāOāŗü[āvé╠āxāōā`āāü[ōŖÄæē’ÄąüAālāIāvāēābāNāXé¬ŹÓ¢▒ōIōŖÄæÄęéŲéĄé─ē┴éĒéķüB
āOāēāNā\üAāuāŖāXāgāŗé╠HIVÄæÄYéĵōŠ
ēpÉ╗¢“æÕÄĶāOāēāNā\üEāXā~āXāNāēāCāōé═üAĢ─ō»ŗŲāuāŖāXāgāŗüEā}āCāäü[āYüEāXāNāCāué¬ŖJöŁÆåé╠łĻśAé╠HIVÄĪŚ├¢“éŹ┼æÕ15ēŁāhāŗüi¢±1800ēŁē~üjé┼ĵōŠéĘéķüBŗŲÉčé╠ŹDÆ▓é╚Ģ¬¢ņé╔éóé┴é╗鿌═éō³éĻéķŹlé”éŠüB
āOāēāNā\é═é▄éĖ3ēŁ1700¢£āhāŗéÄxĢźéóüAÄĪī▒鬏┼ÅIÆiŖKé╔éĀéķHIVÄĪŚ├¢“éĵōŠéĘéķüBīŃé╔ŖJöŁē▀Æ÷éŌ¢┌ĢWÆBɼé╔ē×éČüAā}āCāŗāXāgü[āōāyāCāüāōāgéŲéĄé─5ēŁ1800¢£āhāŗéÄxĢźéżē┬ö\ɽé¬éĀéķüB
ÄĪī▒é¬ÅēŖ·ÆiŖKé╔éĀéķHIVÄĪŚ├¢“é╠ā|ü[āgātāHāŖāIé╔é═é▄éĖ3300¢£āhāŗéÄxĢźéżüBā}āCāŗāXāgü[āōāyāCāüāōāgéŲéĄé─ÆŪē┴éĘéķē┬ö\ɽé¬éĀéķé╠é═5ēŁ8700¢£āhāŗéŠüB
ŹĪē±é╠ĵł°é┼üAāOāēāNā\é═ē▀ö╝ÉöŖöéĢ█ŚLéĘéķHIVÄ¢ŗŲé╠ŹćĢ┘ē’ÄąüAViiVāwāŗāXāPāAé╠āpāCāvāēāCāōéŖgÅ[é┼é½éķéµéżé╔é╚éķüBViiVé╠ÅŁÉöŖöé═Ģ─ātā@āCāUü[éŲē¢¢ņŗ`É╗¢“é¬Ģ█ŚLéĄé─éóéķüBViiVé═HIVÄĪŚ├¢“é╠ÄsÅĻāVāFāAé┼Ģ─āoāCāIłŃ¢“Ģiē’Äąé╠āMāŖāAāhüEāTāCāGāōāVāYé╔Ĥé«2ł╩üB
18 December 2015
GSKüfs global HIV business ViiV Healthcare to acquire Bristol-Myers Squibbüfs R&D HIV assets
GlaxoSmithKline plc today announced that its global HIV business, ViiV Healthcare, has reached two separate agreements with Bristol-Myers Squibb, to acquire its late-stage HIV R&D assets; and to acquire Bristol-Myers Squibbüfs portfolio of preclinical and discovery stage HIV research assets.
Under the terms agreed in the two transactions, ViiV Healthcare will acquire:
These potential therapies have novel modes of action and would offer significant new treatment options to patients with HIV. In addition to being developed as standalone treatment options, these new assets complement ViiV Healthcareüfs existing portfolio and therefore offer multiple opportunities for development as combination therapies.
The late-stage asset purchase comprises an upfront payment of $317 million, followed by development and first commercial sale milestones of up to $518M, and tiered royalties on sales.
The purchase of preclinical and discovery stage research assets comprises an upfront payment of $33 million, followed by development and first commercial sales milestones of up to $587M, and further consideration contingent on future sales performance.
David Redfern, Chief Strategy Officer at GSK and Chairman of ViiV Healthcare, said: ügThese acquisitions strengthen our leadership and innovation in HIV, one of our core areas of scientific research at GSK. The addition of two potential first-in-class late-stage treatments and several promising early clinical development programmes strengthens ViiV Healthcareüfs pipeline and provides us with further new opportunities for growth.üh
The two transactions are anticipated to complete independently during the first half of 2016, subject to necessary approvals, anti-trust and regulatory clearances.
GSK has been a leader in research and development of HIV since the 1980s, with the development of the worldüfs first breakthrough medicine for HIV patients, Retrovir (zidovudine). Development of new treatment options has continued, with Tivicay (dolutegravir) and Triumeq (dolutegravir/ abacavir/lamivudine), the most recent successful innovation. Research programmes are ongoing to develop new treatments and improve drug regimens, including the investigational long-acting integrase inhibitor, cabotegravir. Established in 2009, ViiV Healthcare is a specialist global HIV company, majority owned by GSK, with Pfizer and Shionogi as other shareholders.
GSK – one of the worldüfs leading research-based pharmaceutical and healthcare companies – is committed to improving the quality of human life by enabling people to do more, feel better and live longer. For further information please visit www.gsk.com.
GlaxoSmithKline to 'drop patents in poor
countries for better drug access'
Pharmaceutical firm GlaxoSmithKline has said it wants to make it easier for
manufacturers in the world's poorest countries to copy its medicines.
The British company said it would not file patents in these countries.
Chief executive Sir Andrew Witty said he wanted to take a "graduated" approach
to the company's "intellectual property" based on the wealth of nations around
the globe.
Experts have described the plans as "brave and positive".
GSK hopes that by removing any fear of it filing for patent protection in poorer
countries it will allow independent companies to make and sell versions of its
drugs in those areas, thereby widening the public access to them.
'Clear and simple'
Sir Andrew said he hoped Africa would benefit most from the move.
In accordance with international guidelines set out by the United Nations and
World Bank, the company has drawn up a list of 50
countries with a combined population of about 1 billion people, where it
has said it will not file for patents.
In what GSK describes as lower middle income countries
it will continue to file patents, but will grant licences to generic
manufacturers in exchange for a "small royalty".
Sir Andrew added: "The changes we are setting out aim to make it as clear and
simple as possible for generic manufacturers to make and supply versions of GSK
medicines."
The company has said it also wants to put all its future cancer drugs into a
Medicines Patent Pool in an effort to address what
it described as "the increasing burden of cancer in developing countries".
łŃ¢“Ģiō┴ŗ¢āvü[āŗüiThe Medicines Patent PoolüAMPPüjé═üAHIVÄĪŚ├éųé╠āAāNāZāXéŹéé▀ÉóŖEōIé╔ÉVéĄéóāCāmāxü[āVāćāōéæŻéĘéĮé▀üAUNITAIDé╔éµé┴é─2010öNé╔ænÉ▌é│éĻéĮŹæśAÄxēćægÉDé┼éĀéķüBō»ŹÓÆcé═Ŗ∙æČłŃ¢“Ģié╠ÆßāRāXāgö┼üiāWāFālāŖābāN¢“üjé╠É╗æóé╔ī³é»é─üAō┴ŗ¢āēāCāZāōāXéīÅ┬éĘéķéŲéóéżÉVéĄéóīÆŹNāAāvāŹü[ā`éƱŗ¤éĄüAāüü[āJü[éŲŗ”Ś═éĄé─Äqŗ¤é╔ōKéĄéĮæĮŹ▄Ź¼Źć¢“üiFDCüjé©éµéčÉ╗Ź▄é╚éŪüAĢKŚvéŲéĘéķÉVéĄéóāeāNāmāŹāWü[é╠ŖJöŁéŦŚŃéĘéķüBMPPé═ŹĪō·é▄é┼é╔üAłŃ¢“Ģiāüü[āJü[é¬ō»ægÉDé®éńāēāCāZāōāXéľé»éĮ8ÄĒé╠ŹRāīāgāŹāEāCāŗāXéŲ6ÄĒé╠āWāFālāŖābāN¢“é┼āēāCāZāōāXī_¢±éīŗé±é┼éóéķüB
łŃ¢“Ģiō┴ŗ¢āvü[āŗüiThe Medicines Patent PoolüAMPPüjéŲViiV Healthcare é═2014öN4īÄ2ō·üAHIVÄĪŚ├¢“é╔ŖųéĘéķÉVéĮé╚āRāēā{āīü[āVāćāōéöŁĢ\éĄüAɼÉlé©éµéčżÄֳь├ī³é»é╠ÉVéĄéóŚL¢]é╚ŹRāīāgāŹāEāCāŗāX¢“üiARVüjüAāhāŗāeāOāēārāŗüidolutegravirüADTGüjéųé╠āAāNāZāXŗŁē╗é╔æ╬éĘéķ2é┬é╠ī_¢±éīŗé±éŠüB
ī_¢±é═2013öNé╔öŁĢ\é│éĻéĮMPP-ViiVƱīgŖųīWé╔ŖŅé├éóé─éóéķüBī╗Źsé╠ƱīgŖųīWé═üAżÄÖARVé©éµéčÉóŖEĢ█īÆŗ@ŖųüiWHOüjÉäŦé╔éµéķÄqŗ¤ī³é»é╠ÄÕéĮéķŹRāīāgāŹāEāCāŗāX¢“é╔ŖųéĘéķŹLö═é╚āRāēā{āīü[āVāćāōé¬Ŗ▄é▄éĻéķüBViiVé═MPPé╔æ╬éĄé─üAāhāŗāeāOāēārāŗüiDTGüjé╔æ╬éĘéķ2é┬é╠āēāCāZāōāXéŗ¤Ś^éĘéķüBé╗é╠1é┬é═ɼÉlī³é»é╔ō»ÄĪŚ├¢“éÉ╗æóéĘéķé▒éŲüAéÓéż1é┬é═żÄÖŚpé╠é╗éĻé┼éĀéķüB2é┬é╠āēāCāZāōāXé╔éµé┴é─ÉóŖEé╠éŪé▒é╔éĀé┴é─éÓāWāFālāŖābāN¢“āüü[āJü[é═üAāAāoāJārāŗüiABCüjéŖ▄é▀é─é╗é╠æ╝é╠āWāFālāŖābāN¢“éDTGéŲŹ¼ŹćéĘéķīĀŚśéŲéŲéÓé╔üA1ē±é╠Ģ׌pī`Ä«é┼2é┬éÓéĄéŁé═é╗éĻł╚ÅŃé╠ŖłÉ½łŃ¢“ĢiéŲé╚éķæĮŹ▄Ź¼Źć¢“üiFDCüjéŖJöŁéĘéķīĀŚśéŚ^é”éńéĻéķüB
The patents pool was established in 2010 and
has proved successful in accelerating access to treatments such as HIV,
tuberculosis and hepatitis C through voluntary licensing arrangements, which
allow generic versions of GSK's drugs to be made and distributed in poorer
countries.
Expanding the pool to include cancer drugs will "add to the wider contribution
GSK makes to improve access to effective healthcare around the world", the
company said.
Sir Andrew added: "The experience GSK has with the Medicines Patent Pool for
Tivicay - our newest HIV medicine and one of our
most commercially successful products - gives us confidence that increasing
access, incentivising innovation appropriately and achieving business success
can go hand in hand."
GSK said it would continue to seek full patent protection in richer parts of the
world.
'Broadening access'
Prof Raymond Hill, former President of the British Pharmacological Society, said
GSK's plans set a precedent for other major pharmaceutical companies to follow.
He said: "This is a brave and positive step towards broadening the access to
important new medicines in the developing world.
"The impact of this move on the treatment of cancer and other diseases in each
individual country will depend on whether there is a local adequate healthcare
infrastructure that will allow the safe use of powerful new drugs in an
appropriate group of patients.
"Many new cancer drugs are most valuable when used in sub-groups of patients
identified by advanced diagnostic techniques that may not be available."
Prof Alan Boyd, from the Royal Colleges of Physicians, described the plans as
"good news" and "significant"
He added: "Access to medicines for patients on a global basis is vital and it is
good to see an innovative approach like this to ensure this happens."
--------
Building on this perspective, GSK is evolving its graduated approach to filing and enforcing patents so that IP protection reflects a countryüfs economic maturity.
ü@

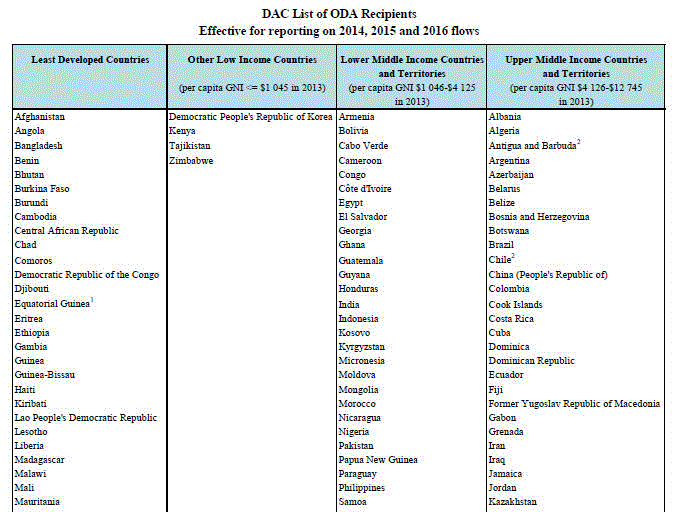
ŹæśAü@ŖJöŁēćÅĢłŽł§ē’ üiDAC)

ü[ü[ü[
Novartisé═2014öN4īÄ22ō·üAæÕŗK¢═é╚Ä¢ŗŲŹ─ĢęéöŁĢ\éĄéĮüB
GlaxoSmithKline (GSK) é®éńŹRé¬é±Ź▄É╗ĢiīQéöāĹéĘéķéŲéŲéÓé╔üAæÕÅO¢“Ä¢ŗŲé═GSKé╠Ä¢ŗŲéŲōØŹćéĄüAGSKÄÕæ╠é╠JVéŲéĘéķüBŹXé╔āCāōātāŗāGāōāUł╚ŖOé╠āÅāNā`āōÄ¢ŗŲéGSKé╔öäŗpéĘéķüB
é▒éĻéŲé═Ģ╩é╔āCāōātāŗāGāōāUāÅāNā`āōé╠öäŗpéÉié▀é─é©éĶüAō«Ģ©¢“Ä¢ŗŲé═Eli Lillyé╔öäŗpéĘéķüBé▒éĻé╔éµéĶüANovartisé═Ä¢ŗŲéłŃ¢“ĢiüAEye careÉ╗ĢiüAGenerics é╠3é┬é╔ŹiéĶŹ×é▐üB
Novartisé╠æÕÅO¢“üiOTC)Ä¢ŗŲéŲGSKé╠Consumer HealthcareéōØŹćéĄüAJVéÉ▌Ś¦éĘéķüB
JVé═öäÅŃŹé鬢±100ēŁāhāŗé┼üAÆåÉSéŲé╚éķ4Ģ¬¢ņüAīÆŹNüAī¹ŹoēqÉČüAēhŚ{üAöńĢåé╠īÆŹNé╔é©éóé─ÄÕō▒ōIÆnł╩éÉĶé▀éķüB
ɵÉiŹæé╠é▌é┼é╚éŁüAāuāēāWāŗüAÆåŹæüAāüāLāVāRüAāŹāVāAōÖé╠ɼÆĘÆśéĄéóōrÅŃŹæé╔éÓŗÆō_éÄØé┬üB
Novartisé═é▒é╠JVé╔36.5%éÅoÄæéĄüAÄsÅĻŖöē┐éŖŅÅĆé╔éĘéķÄ¢æOé╔īłé▀éńéĻéĮĢ¹Ä«é╔ŖŅé├éŁŖöē┐é┼ÄØŖöéöäŗpéĘéķīĀŚśéÄØé┬üB2014/4/25ü@NovartisüAGSKéŲé╠ĵł°ōÖé┼Ä¢ŗŲŹ─Ģęü@
Horlicks é═üAĢ▓¢¢ö×ēĶł∙Ś┐é╠āuāēāōāhüB ÅAÉQæOé╔ł∙é▐éŲÉć¢░éæŻéĄüAł└¢░é┼é½éķéŲé│éĻéķüBŗīāCāMāŖāXÉA¢»Æné┼ā|āsāģāēü[é╚ł∙é▌Ģ©üB
ü[ü[ü[
GlaxoSmithKline plc today announces that it has reached an agreement with Novartis for the buyout of Novartisüf 36.5% stake in their Consumer Healthcare Joint Venture for $13 billion (£9.2 billion). The Consumer Healthcare Joint Venture was formed as part of the three-part transaction between GSK and Novartis which was approved by shareholders in 2014. Last year, GSKüfs Consumer Healthcare business reported sales of £7.8 billion and since 2015 sales have grown 4% on a 3 year CAGR basis (2015-2017 at 2014 CER) with an overall improvement in operating margins from 11.3% in 2015 to 17.7% in 2017.
Under the terms of the original transaction, Novartis has the right, exercisable from 2 March 2018 to 2 March 2035 to require GSK to purchase its stake (or specified tranches of it) in the Joint Venture. This put option, in both size and possible timing, creates inherent uncertainty for the Groupüfs capital planning. The new agreement to buy-out Novartisüf stake removes this uncertainty and improves the Groupüfs ability to plan allocation of capital to its other priorities.
As a result of the transaction, GSKüfs shareholders will capture the full value of GSKüfs Consumer Healthcare growth. With category-leading Power Brands, increased focus on science-based innovation and improved operational efficiencies, GSK Consumer Healthcare is well positioned to deliver sales growth, operating margin improvements and attractive returns. The business expects operating margins to approach üemid-20üfsüf percentages by 2022 at 2017 CER.
The transaction is expected to be accretive to adjusted earnings in 2018 and thereafter, and is expected to strengthen operational cash flows. Together with the Groupüfs new launch opportunities and expected operational improvements, these financial benefits further support GSKüfs increased confidence in its ability to deliver its 2020 outlooks and invest effectively in the Groupüfs other priorities.
The transaction is subject to approval by GSK shareholders as Novartis is treated as a related party under UK Listing Rules, and the Board intends to unanimously recommend that shareholders vote in favour of the transaction.
GSK is initiating a strategic review of Horlicks and its other consumer healthcare nutrition products to support funding of the transaction, and to drive increased focus on OTC and Oral Health categories. Combined sales of these products were approximately £550 million in 2017.
The majority of Horlicks and other nutrition products sales are generated in India, with the Horlicks range widely recognised as a portfolio of premium nutrition products. In India, these products are sold by GlaxoSmithKline Consumer Healthcare Ltd, a public company listed on the National Stock Exchange (NSE) and Bombay Stock Exchange (BSE). The strategic review will include an assessment of GSKüfs 72.5% shareholding in the company.
GSK expects the outcome of the strategic review to be concluded around the end of 2018. There can be no assurance that the review process will result in any transaction.
India remains a priority market for GSK investment and growth. The Consumer Healthcare business will continue to invest in growth opportunities for its OTC and Oral Health brands, such as Sensodyne and Eno. The Group is also actively investing in its Pharmaceutical and Vaccines businesses, including building new manufacturing capacity in Vemgal, Karnataka and Nashik.
Emma Walmsley, Chief Executive Officer, GSK said:
ügThe proposed transaction addresses one of our key capital allocation priorities and will allow GSK shareholders to capture the full value of one of the worldüfs leading Consumer Healthcare businesses. For the Group, the transaction is expected to benefit adjusted earnings and cash flows, helping us accelerate efforts to improve performance. Most importantly it also removes uncertainty and allows us to plan use of our capital for other priorities, especially pharmaceuticals R&D.üh
ü@
ü@